Earlier research of AD Chemicals already showed that etching plays an important role in increasing the quality of pre-treatment of recycled aluminium. This is why the next step of the research involved tests on recycled aluminium while taking one alteration into account: the etching process. Multiple samples were researched and are displayed in Table 2: Extrusion alloy examples. From Table 2 can be concluded that the testing materials include a maximum permitted amount of copper. Besides this, the Zinc value is not 1:1. This testing method is frequently used by extruders to compensate the negative effects of a different alloy composition.

Table 2: Extrusion alloy examples
The results of this test (after 1000 hours AAST) can be seen in an overview in image 3. This shows that Cleaner 502, the regular etching product, from AD Chemicals performs just above the standard. With the first optimization step (For example in Cleaner 602 min. See also Image 4: Testing results of optimized etch) a clear improvement is already visible and with the fully optimized product (Cleaner 602) a perfect result is achieved.
The improvement was realized because AD Chemicals developed an additive which can be added to an etching bath. This additive improves the rinse-ability of the substrate. The rinse-ability is important because a.o. increased Zinc- and Copper values in the bath, as a result of intensive etching (Seaside), can result in precipitation of these elements on aluminium. This effect is comparable with the situation where aluminium and Zinc parts are treated in the same pre-treatment, which also results in Zinc precipitation on aluminium.
Furthermore, it is important to mention that the conversion layer itself plays no role in the optimization process, and has no effect on the results.
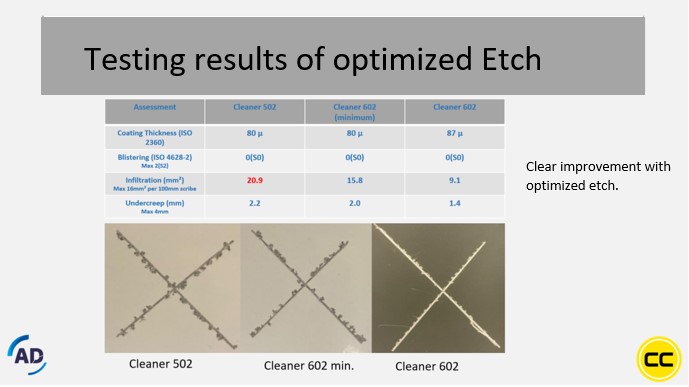
Image 4: Testing results of optimized etch
Testing methods (filiform) Corrosion
Qualicoat Belgium has been working for some time according to specification §2.19, which advices an adjusted testing method, whereby the tested parts are exposed along the cutting edge to hydrochloric acid by means of dripping for one minute. After which they are placed in a climate cabinet for a 1000 hours at a temperature of 40 degrees and a humidity air exposure of 82%. However the performance of this test can differ per test executive. For example, even the amount of drops to use, how to dab, where to dab with etc. can make a big difference.
A cross check among various internal and external laboratories shows that the same test samples do not always produce the same results. Due to this, it is possible that two of three samples achieve a 100% and 1 fails, even though they have been treated and tested identically.
This does not mean the method is not functioning, but it does imply that this new method is more sensitive than the vapor test that was applied previously according to ISO4623.
Another important aspect to consider is that a powder coating is permeable. The hydrochloric acid can affect the coating. If a drop of hydrochloric acid falls on the paint, “filiform effects” can be observed on that part of the coating. If the coating is hit during the process of dripping into the scratch, this can have an effect on the outcome of the test.
Based on these aspects it becomes clear that various conditions in the supply chain are important for achieving a qualitative final result. Changed conditions in the design phase, the coating process, the construction phase, and the testing phase, can all affect the final product quality. AD Chemicals and Compri Coating Service are happy to discuss these developments with interested companies and institutes, such as aluminium extruders, laboratories, and coating manufacturers. It is their vision that by good collaboration in the full supply chain everyone will be ready for today’s and tomorrow’s challenges.
Innovation: PreCoat F31/20 (D)
In addition to the above mentioned optimization in the etching process, there are other ways to cope with the developments in the market. For example, PreCoat F31/20(D) of AD Chemicals makes it possible to achieve an excellent corrosion protection without etching. The product is based on high-end state-of-the-art aerospace technology. The uniqueness of the product lies in the fact that it is a complete chemical pre-treatment that can be applied in just one treatment step. PreCoat F31/20(D) can potentially replace a full pre-treatment line by a simple process without heated baths and the use of dangerous chemicals. Implementing this process will depend on how clean your aluminium is. In some cases a degreasing would be necessary but the etching bath can always be eliminated. This can result in a significant reduction of the CO2 footprint for many applicators. The achieved testing results can be found in Image 5: Testing results PreCoat F31/20 (D). Even the CASS test, a severe corrosion test commonly used in the automotive industry, was passed successfully. This novel pre-treatment method is already widely embraced in i.e. the truck and trailer and agriculture industry.
However, etching is a requirement according to the Qualicoat standard and this new product is therefore currently not yet an option for Qualicoat applicators. AD Chemicals is open for discussion with Qualicoat “because innovation can only be achieved by thinking outside-the-box” says Roland van Meer, AD Chemicals.“

Extensive production experience Compri Coating Service
Compri Coating Service already has more than half a year of production experience with the optimized etching process. Ludo Appels of Compri Coating Service about their experience: “We as applicator are well prepared with the optimized etching process for the growing volume of recycled aluminium. Robustness is essential in our process because we, as an applicator, do not always know which type of aluminium needs to be coated. With this optimized process we are able to offer our customers the highest coating quality for their demands on recycled aluminium”. This process has been implemented throughout the entire Compri Coating Group. AD Chemicals and Compri Coating Services are open for discussion and are happy to share knowledge with interested institutes, extruders or applicators. Together for a strong and futureproof supply chain with recycled aluminium!